AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Atomic size trend ap che1/14/2024  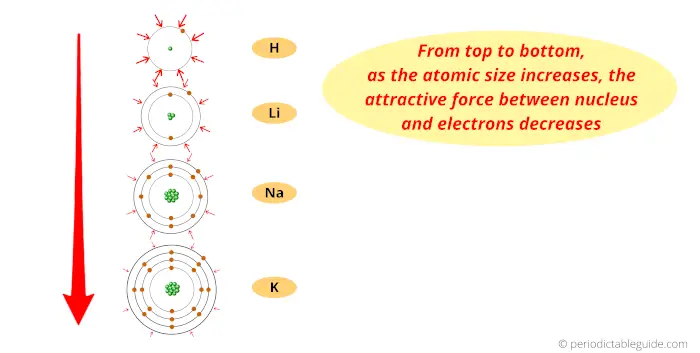 
the pair of dots represent valence electrons that are not involved in bonding, called lone pair of electrons.Each line in these molecules represents a bonding electron pair, and Shells 5 through 7: 32 electrons in any known element, however there are additional orbitals available to hold even more electrons, but there is no element with a large enough atomic number to fill up those slots.Carbon, nitrogen, oxygen, and fluorine with 4, 3, 2, and 1 unpaired dot can make 4, 3, 2, and 1 bond, e.g., in the following molecules:, ,, and. A bond is formed by sharing unpaired valence electrons. A bond is represented by a line between the bonded atoms. Generally, each unpaired dot can make one bond.įor example, a hydrogen atom with one unaired dot can make one bond as in H-H. AP Chemistry: Periodic Table, Chapter 2 - Periodic Trends Term 1 / 55 Atomic Radii Click the card to flip Definition 1 / 55 A periodic trend which increases from the top to the bottom of a group, because each increase involves another larger, energy level. The electron dots in the Lewis structure are a convenient way to determine how many bonds an atom of an element can make. It increases as you move from left to right across the periodic table. 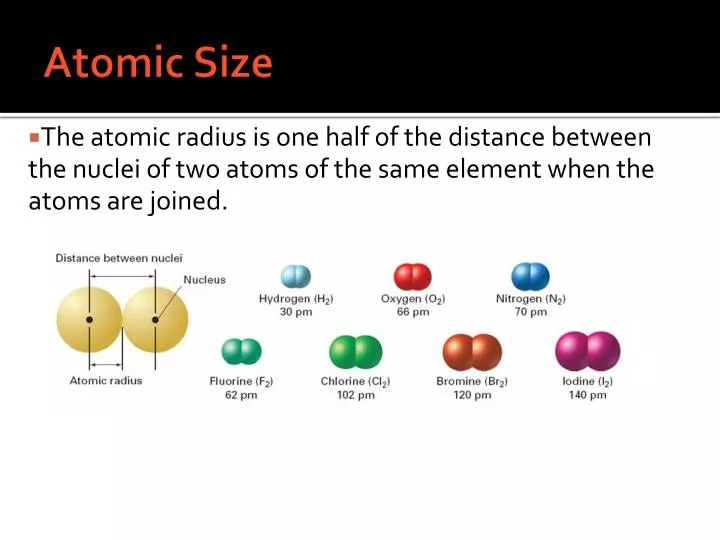
Electrons not in the valence shell shield the valence electrons from the feeling the full effect of the nucleus. Thus, it takes much more energy than just overcoming a larger ionic charge would suggest.\): Lewis symbols or electron-dot symbols of the first twenty elements in the periodic table. The charge of the nucleus felt by the valence electrons. The radius increases as you move down a group (column) and decreases as you move from left to right across a period (row). Why is it so much larger? Because the first two electrons are removed from the 3 s subshell, but the third electron has to be removed from the n = 2 shell (specifically, the 2 p subshell, which is lower in energy than the n = 3 shell). Both atomic radius and ionic radius follow a trend on the periodic table. Compare electron affinities and electronegativities. 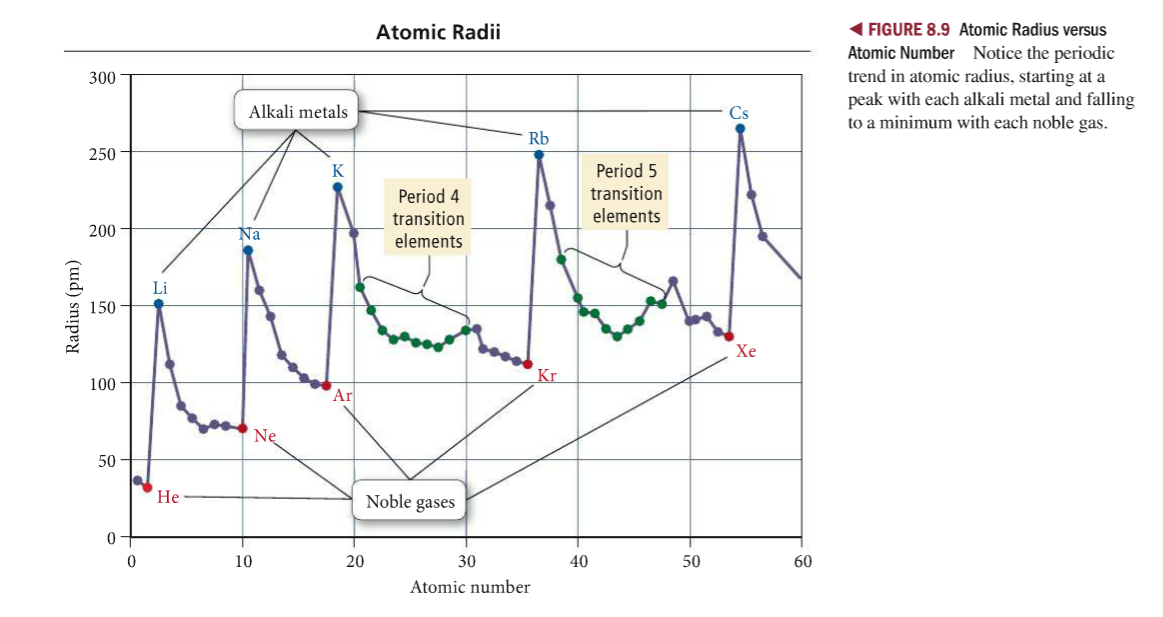
K has one electron in the fouth energy level and Kr has. The differences in atomic radii are the result of differing amounts of protons between atoms whose electrons are in the same energy level. Create An Account Create Tests & Flashcards. The third IE, however, is over five times the previous one. Predict greater or smaller atomic size and radial distribution in neutral atoms and ions. AP Chemistry : Periodic Trends Study concepts, example questions & explanations for AP Chemistry. And so we start with hydrogen, which has atomic number of 1, which means that it has one proton in the nucleus. And let's go ahead and sketch out the atoms first. 32.Typically form ions with charges of +3 and +6 Use the following responses to answer questions 33-37. Show a reversal in the trend for first ionization energy because of shielding by full orbitals. And so here we have two elements found in group one, so hydrogen and lithium. Show a reversal in the trend for first ionization energy because of electron-electron repulsions. The second IE is twice the first, which is not a surprise: the first IE involves removing an electron from a neutral atom, while the second one involves removing an electron from a positive ion. Let's look at the trends for atomic radius, and first we'll start with group trends. 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
